Sheep Diseases
Contagious Respiratory Tumours
Also known as: Jaagsiekte, Pulmonary Adenomatosis and Adenomatosis – Pulmonary

Breed does not appear to influence the incidence or outcome of contagious respiratory tumour. Unfortunately, this fatal disease is present in most sheep rearing countries worldwide.
There are two neoplastic conditions which result in contagious respiratory tumours (or contagious lung cancer) in small ruminants:
- Ovine Pulmonary Adenocarcinoma (also known as ODA, Sheep Pulmonary Adenomatosis, SPA, Jaagsiekte, pulmonary carcinoma)
- Enzootic Nasal Adenocarcnoma (also referred to as ENA, Enzootic Nasal Tumour)
Both conditions are highly contagious, have major welfare implications and are caused by two distinct but closely related betaretroviruses. OPA is caused by the oncogenic betaretrovirus, jaagsiekte sheep retrovirus (JSRV) (De las Heras et al., 2003), and ENA is caused by Enzootic Nasal Tumour Virus (ENTV). OPA affects mainly sheep, but there are occasional reports in goats (Sharp and De las Heras, 2007). ENA affects both sheep and goats, sub-species ENTV-1 infects sheep, and ENTV-2 infects goats.
OPA is a problem to the sheep industry worldwide, except in Australia, New Zealand and Iceland (Cousens et al., 2015). ENA is also a global issue but appears to be absent in Britain, Australia and New Zealand (Sharp and De las Heras, 2007). In both conditions tumours arise from secretory epithelial cells that retain their secretory function after transformation (Salvatori et al., 2004). It is thought the tumour tissue reactivates latent herpesvirus as a result of immunosuppression (Myer et al., 1988).
Ovine Pulmonary Adenocarcinoma
Recently it has been demonstrated that age at infection has an effect on the development of OPA, and that resistance to disease increases with age (Salvatori et al., 2004). The most common age bracket of sheep to show clinical signs is 3-4 years of age (Scott et al., 2013), when seen in sheep younger than this, it may be as progeny of a dam with advanced clinical disease.
Clinical signs are those of progressive respiratory illness, exercise intolerance, loss of weight despite good appetite and often overproduction of fluid in the lungs which may flow from the nostrils when the head is lowered (De las Heras et al., 2003). Some experts recommend the wheelbarrow test (to test for the disease, however it is not definitive). Respiratory problems are most obvious after exercise, often associated with noticeable movement of the abdominal wall and high-pitched and moist sounds. Death from ODA inevitably occurs, often suddenly, and frequently complicated by secondary bacterial infection such as pasteurella haemolytica pneumonia (Cousens et al., 2015; Myer et al., 1988).
Elevate the hind quarters above the head and chest; leakage of between a few millimetres and up to half a litre of clear white, or frothy virus-laden fluid pours from the nostrils.
This method is not definitive in diagnosing ODA infection as unfortunately early cases do not appear to produce enough fluid to be detected by the wheelbarrow test, and even in severely infected sheep, production of fluid is inconsistent (Cousens et al., 2015).
Enzootic Nasal Adenomatosis
In ENA, tumours can arise in the nose bilaterally, or unilaterally. ENA is contagious and spread via the respiratory secretions. Clinical signs include copious nasal secretions, open mouthed breathing, dyspnoea and facial asymmetry (Walsh et al., 2013).
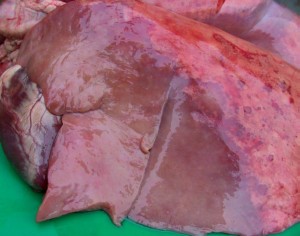
Ovine Pulmonary Adenocarcinoma causes a virus-induced tumour, which is clearly distinguishable from the rest of the lung tissues (the grey / purple zone). This image is from www.bushhousevetgroup.co.uk
How are Contagious Respiratory Tumours Spread?
Contagious respiratory tumours are transmissible via the respiratory route (Sharp et al., 1983), by coughing, sneezing and perhaps breathing near susceptible sheep. It is likely that infected animals excrete the virus long before they show clinical signs. Housing sheep, together with trough feeding and watering, will increase the spread of the disease. Bought-in sheep are usually the source of infection.
Control and Prevention of Contagious Respiratory Tumours
Control is difficult as early detection is essential in preventing the spread of the disease within and between flocks (Cousens et al., 2015). However, an effective method for controlling the spread of JSRV or ENAV has yet to be developed. The absence of an immunological response to JSRV in infected animals makes it difficult to devise an effective diagnostic test. There is a PCR test available that is highly sensitive in lab tests, but consistently fails to pick up cases in the field other than late stage, overtly clinical presentations. Due to this, and the fact that there is no treatment for contagious respiratory tumours, control should be based on regular inspection of adult sheep in affected flocks. In closed flocks the chances of the flock becoming infected are lower. Any sheep showing loss of weight and respiratory disease should be isolated and veterinary advice sought. The affected animals should be culled immediately to prevent further spread of the disease.
Treating Contagious Respiratory Tumours
There is no treatment for contagious respiratory tumours and affected animals should be humanely destroyed.
Good Practice Based on Current Knowledge
- Maintain a closed flock is the best way to prevent contagious respiratory tumour
In endemic flocks eradication of the disease should be part of the overall health plan:
- Animals should be regularly inspected and any suspected cases isolated
- Veterinary advice should be sought
- Where possible, stocking rates should be lowered and housing avoided to minimise transmission
- Humanely destroy affected animals
- In the case of small flocks it may be better to replace the entire flock
- Source animals from OPA free flocks where possible, but with the absence of suitable commercially available tests this can be difficult


 American English
American English

Comments are closed.